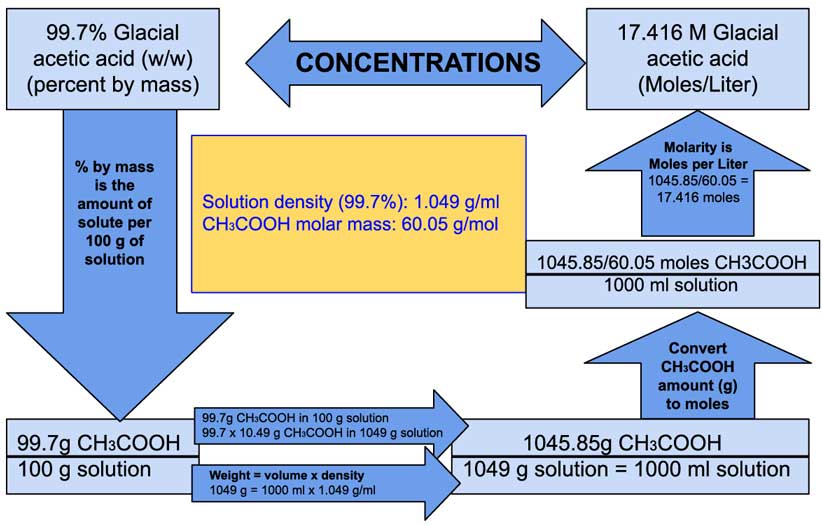
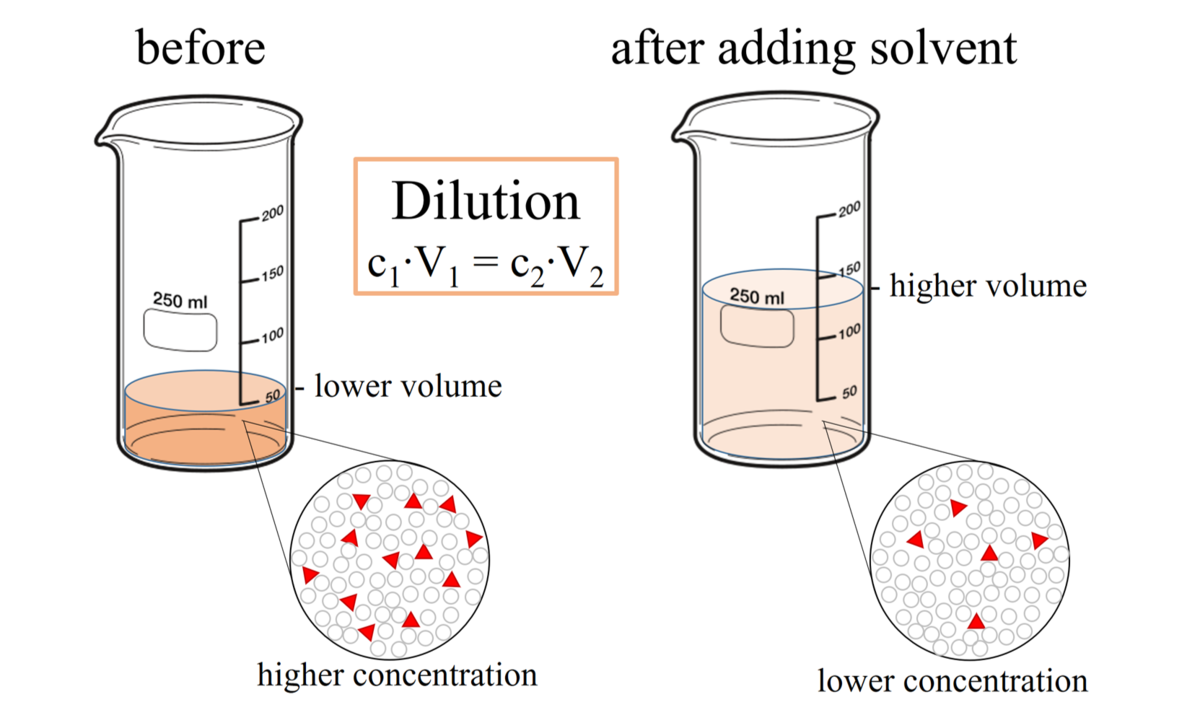
A 0.01 M solution of acetic acid is 1.34 % ionized (degree of dissociation = 0.0134 ) at 298 . What is the ionization constant of acetic acid.
What is the pH of 0.1 m of acetic solution, if acetic acid is a weak acid with Ka2 1.86 × 10-5? - Quora

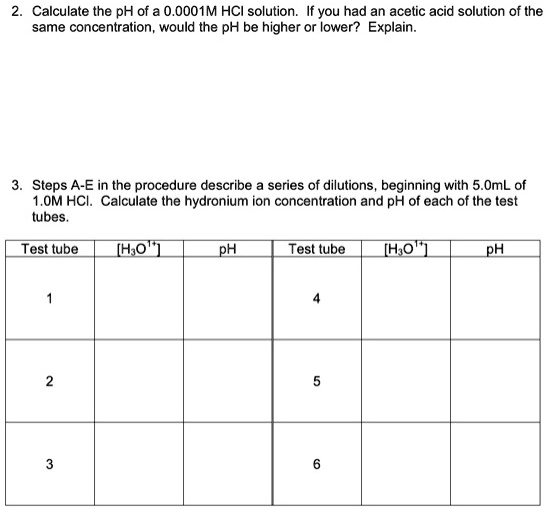
The degree of ionization of a 0.1 M bromoacetic acid solution is 0.132 . Calculate the pH of the solution and the pKa of bromoacetic acid.
How to calculate the pH of 0.010 molarity acetic acid solution, if its dissociation constant is 1.8*10^-5 - Quora

Calculate pH of 100 ml buffer solution containing 0.1 g acetic acid and 0.2 g sodium actetate. | pH calculations and more in fundamentals of pharmaceutics.
![The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ] The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]](https://d1hhj0t1vdqi7c.cloudfront.net/v1/QXVmVDZfQ29GV1k=/sd/)
The pH of 0.1 M acetic acid solution is closest to[Dissociation constant of the acid, Ka = 1.8 × 10^-5 ]
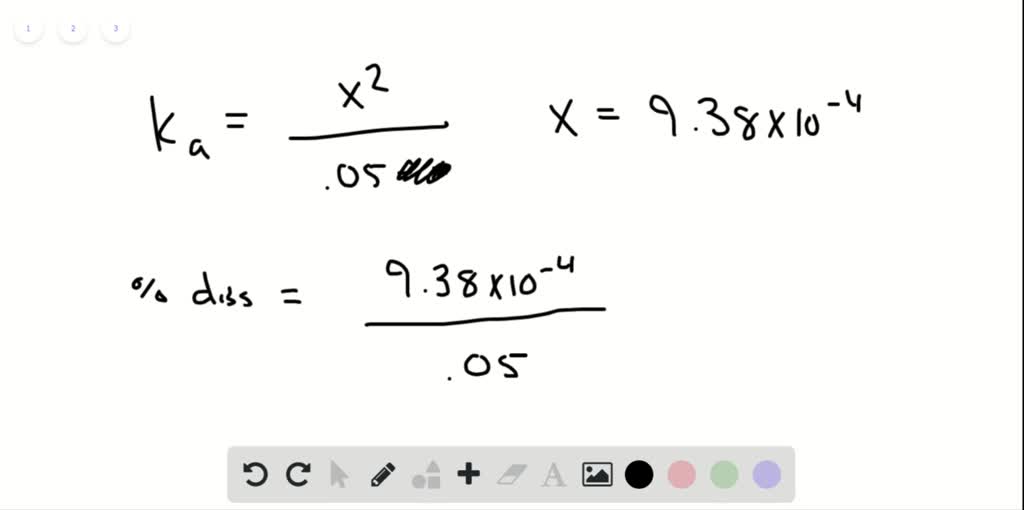
SOLVED:Calculate the percent dissociation of the acid in each of the following solutions. a. 0.50M acetic acid b. 0.050M acetic acid c. 0.0050M acetic acid d. Use Le Châtelier's principle to explain