
PDF) Modelling of the thermophysical properties of citric acid aqueous solutions. Density and viscosity

Production of Nanocellulose Using Citric Acid in a Biorefinery Concept: Effect of the Hydrolysis Reaction Time and Techno-Economic Analysis | Industrial & Engineering Chemistry Research

SOLVED: Molar mass of Citric acid = 190.112 g/mole density of lemon juice =1.00 glml Volume of lemon juice used = 50.00 ml Molarity of NaOH = 1.00 M Vof NaOH used

SciELO - Brasil - Extracion of citric acid in 2-octanol and 2-propanol solutions containing tomac: an equilibria and a LSER model Extracion of citric acid in 2-octanol and 2-propanol solutions containing tomac:
![PDF] A study on viscosities of citric acid and tartaric acid in water and binary aqueous mixtures of ethanol at different temperatures | Semantic Scholar PDF] A study on viscosities of citric acid and tartaric acid in water and binary aqueous mixtures of ethanol at different temperatures | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/5dbb16e7e8bfb291852291795672dbd5092659a1/2-TableI-1.png)
PDF] A study on viscosities of citric acid and tartaric acid in water and binary aqueous mixtures of ethanol at different temperatures | Semantic Scholar

PDF) Densities and Viscosities of Citric Acid in Aqueous Cetrimonium Bromide Solutions with Reference to the Manifestation of Solvation









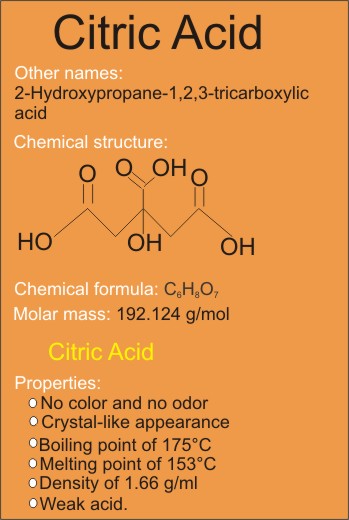

![PDF] Acoustic behaviour of citric acid in aqueous and partial aqueous media | Semantic Scholar PDF] Acoustic behaviour of citric acid in aqueous and partial aqueous media | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6d852008e9b95b4638a5e8010484a5466367bf93/3-Table1-1.png)
