![N-[(2R,5R)-2α,4β-Diphenyl-6-oxo-7-oxa-1-azaspiro[4.4]nonane-3α-yl]-p-toluenesulfonamide | Chemical Substance Information | J-GLOBAL N-[(2R,5R)-2α,4β-Diphenyl-6-oxo-7-oxa-1-azaspiro[4.4]nonane-3α-yl]-p-toluenesulfonamide | Chemical Substance Information | J-GLOBAL](https://jglobal.jst.go.jp/images/chemical/detail/034/492/J3.449.226C.png)
N-[(2R,5R)-2α,4β-Diphenyl-6-oxo-7-oxa-1-azaspiro[4.4]nonane-3α-yl]-p-toluenesulfonamide | Chemical Substance Information | J-GLOBAL
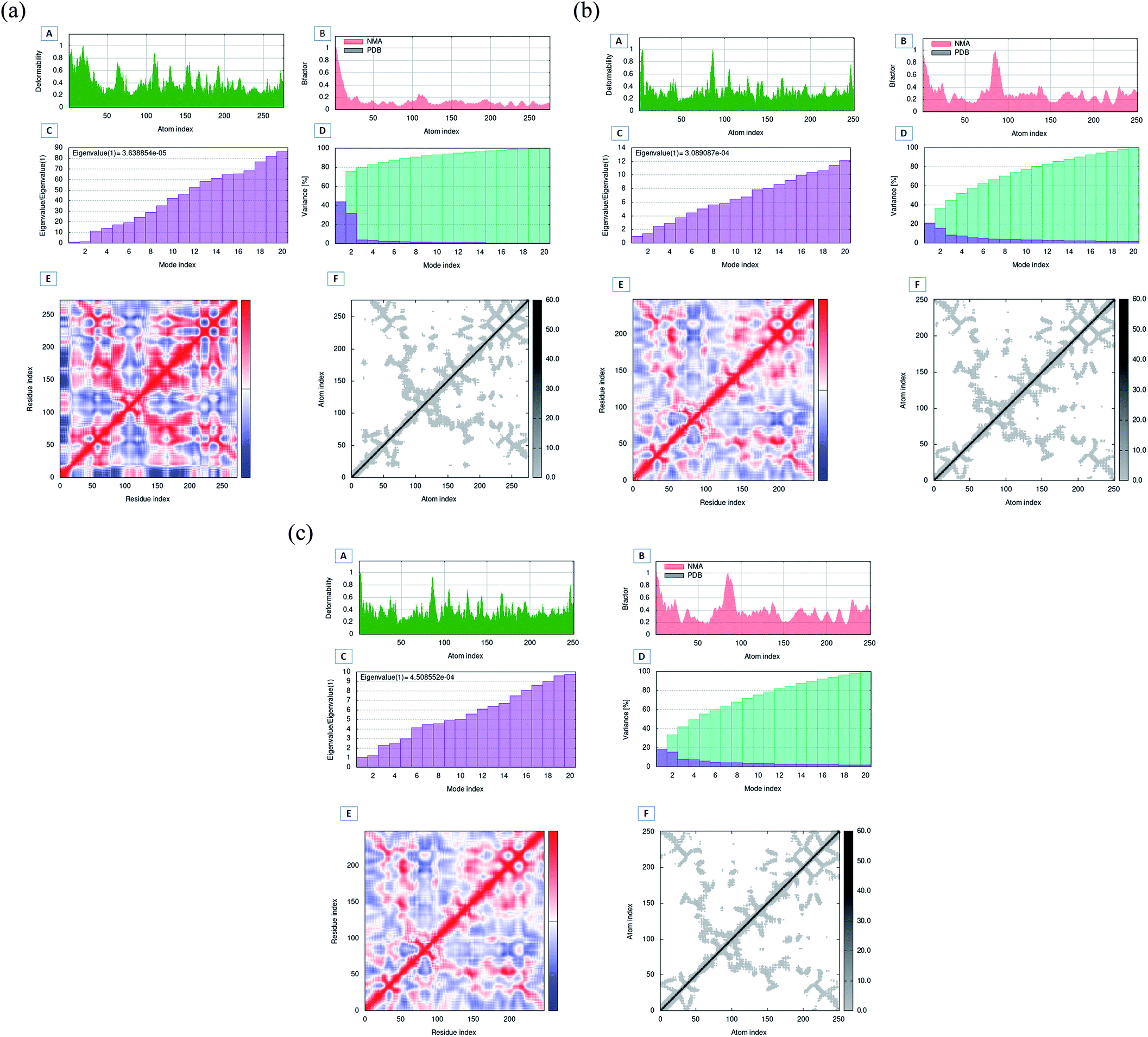
Oxa-376 and Oxa-530 variants of β-lactamase: computational study uncovers potential therapeutic targets of Acinetobacter baumannii - RSC Advances (RSC Publishing) DOI:10.1039/D2RA02939A
![3aS,7aα)-8,8-Dimethyl-3aβ,6β-methanospiro[1-oxa -2-thia(IV)-hydrindane-2,2′-[1-oxa-2-thia(IV)-3]cyclopentene]-5′-one | Chemical Substance Information | J-GLOBAL 3aS,7aα)-8,8-Dimethyl-3aβ,6β-methanospiro[1-oxa -2-thia(IV)-hydrindane-2,2′-[1-oxa-2-thia(IV)-3]cyclopentene]-5′-one | Chemical Substance Information | J-GLOBAL](https://jglobal.jst.go.jp/images/chemical/detail/010/905/J1.090.561C.png)
3aS,7aα)-8,8-Dimethyl-3aβ,6β-methanospiro[1-oxa -2-thia(IV)-hydrindane-2,2′-[1-oxa-2-thia(IV)-3]cyclopentene]-5′-one | Chemical Substance Information | J-GLOBAL
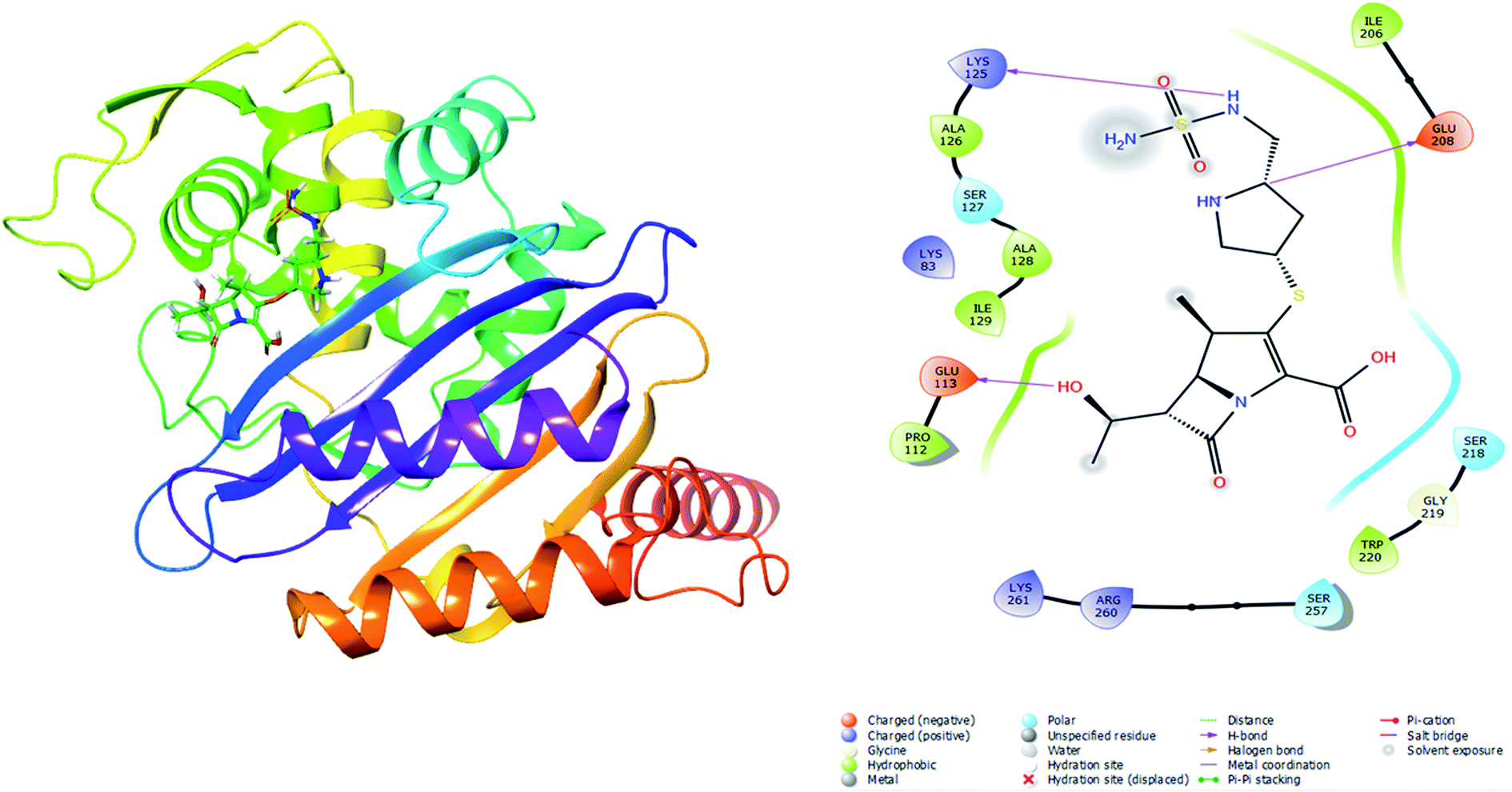
Oxa-376 and Oxa-530 variants of β-lactamase: computational study uncovers potential therapeutic targets of Acinetobacter baumannii - RSC Advances (RSC Publishing) DOI:10.1039/D2RA02939A
![1S,4S,5S,6S)-N,3-Diphenyl-8-methyl-2-oxa-3-phospha(V)-bicyclo[2.2.2]octa-7-ene-5,6-dicarbimide 3-oxide | Chemical Substance Information | J-GLOBAL 1S,4S,5S,6S)-N,3-Diphenyl-8-methyl-2-oxa-3-phospha(V)-bicyclo[2.2.2]octa-7-ene-5,6-dicarbimide 3-oxide | Chemical Substance Information | J-GLOBAL](https://jglobal.jst.go.jp/images/chemical/detail/020/230/J2.023.072J.png)
1S,4S,5S,6S)-N,3-Diphenyl-8-methyl-2-oxa-3-phospha(V)-bicyclo[2.2.2]octa-7-ene-5,6-dicarbimide 3-oxide | Chemical Substance Information | J-GLOBAL

Multiscale Simulations Identify Origins of Differential Carbapenem Hydrolysis by the OXA-48 β-Lactamase | ACS Catalysis
![1S,2R,7S)-8,9-(Methyliminobiscarbonyl)-12-oxa-3,4,8,9-tetraazapentacyclo[4.4.3.2<sup>2,5</sup>.2<sup>7,10</sup>.0<sup>1,6</sup>]heptadecane-3,14-diene 3,4-dioxide | Chemical Substance Information | J-GLOBAL 1S,2R,7S)-8,9-(Methyliminobiscarbonyl)-12-oxa-3,4,8,9-tetraazapentacyclo[4.4.3.2<sup>2,5</sup>.2<sup>7,10</sup>.0<sup>1,6</sup>]heptadecane-3,14-diene 3,4-dioxide | Chemical Substance Information | J-GLOBAL](https://jglobal.jst.go.jp/images/chemical/detail/012/769/J1.276.920B.png)
1S,2R,7S)-8,9-(Methyliminobiscarbonyl)-12-oxa-3,4,8,9-tetraazapentacyclo[4.4.3.2<sup>2,5</sup>.2<sup>7,10</sup>.0<sup>1,6</sup>]heptadecane-3,14-diene 3,4-dioxide | Chemical Substance Information | J-GLOBAL

Programmable Ether Synthesis Enabled by Oxa-Matteson Reaction | Journal of the American Chemical Society
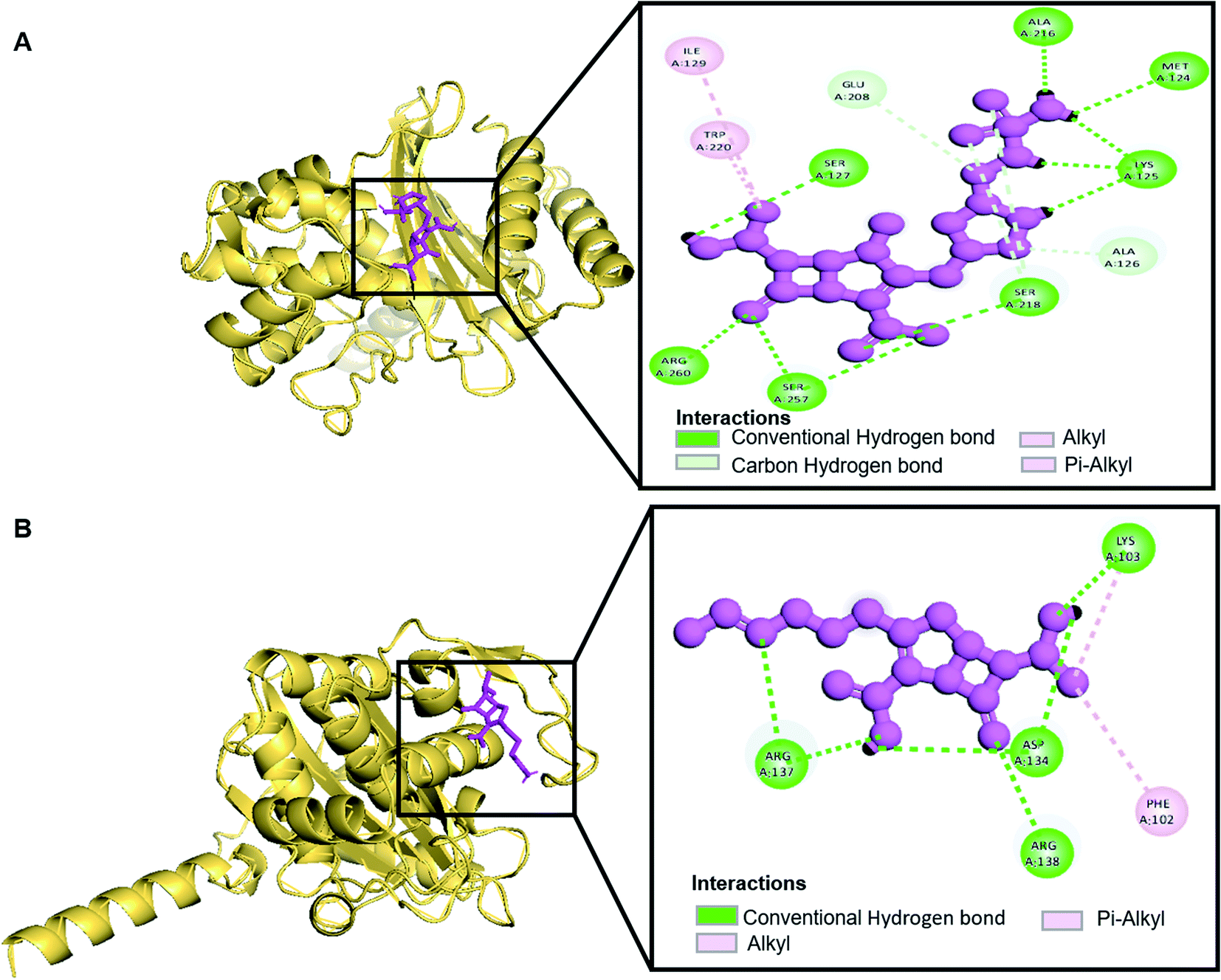
Oxa-376 and Oxa-530 variants of β-lactamase: computational study uncovers potential therapeutic targets of Acinetobacter baumannii - RSC Advances (RSC Publishing) DOI:10.1039/D2RA02939A
![1,3-Dihydroxy-2-[(1-oxa-4-azoniacyclohexane-4-yl)methyl]-4-oxo-1,2,3,4-tetrahydronaphthalene-2-ide | Chemical Substance Information | J-GLOBAL 1,3-Dihydroxy-2-[(1-oxa-4-azoniacyclohexane-4-yl)methyl]-4-oxo-1,2,3,4-tetrahydronaphthalene-2-ide | Chemical Substance Information | J-GLOBAL](https://jglobal.jst.go.jp/images/chemical/detail/020/603/J2.060.308I.png)
1,3-Dihydroxy-2-[(1-oxa-4-azoniacyclohexane-4-yl)methyl]-4-oxo-1,2,3,4-tetrahydronaphthalene-2-ide | Chemical Substance Information | J-GLOBAL

IJMS | Free Full-Text | Specific Amino Acid Substitutions in OXA-51-Type β-Lactamase Enhance Catalytic Activity to a Level Comparable to Carbapenemase OXA-23 and OXA-24/40
![3-Benzyl-2-oxa-3,4-diaza-2H-benzo[f]indene-1(3H)-one | Chemical Substance Information | J-GLOBAL 3-Benzyl-2-oxa-3,4-diaza-2H-benzo[f]indene-1(3H)-one | Chemical Substance Information | J-GLOBAL](https://jglobal.jst.go.jp/images/chemical/detail/034/915/J3.491.512A.png)




![Recombinant Acinetobacter Beta-lactamase OXA-23 Protein [230-00712] Recombinant Acinetobacter Beta-lactamase OXA-23 Protein [230-00712]](https://www.raybiotech.com/images/thumbnails/280/350/detailed/63/230-00712.jpg)
![Oxa[7]helicenen | Chemical Substance Information | J-GLOBAL Oxa[7]helicenen | Chemical Substance Information | J-GLOBAL](https://jglobal.jst.go.jp/images/chemical/detail/027/878/J2.787.830J.png)