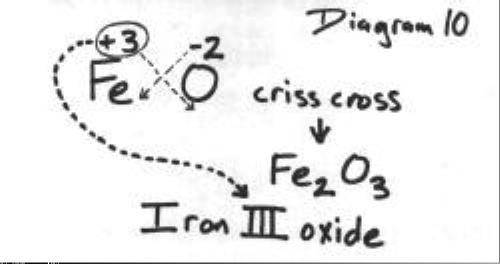
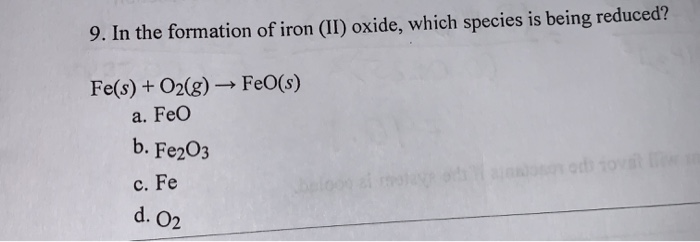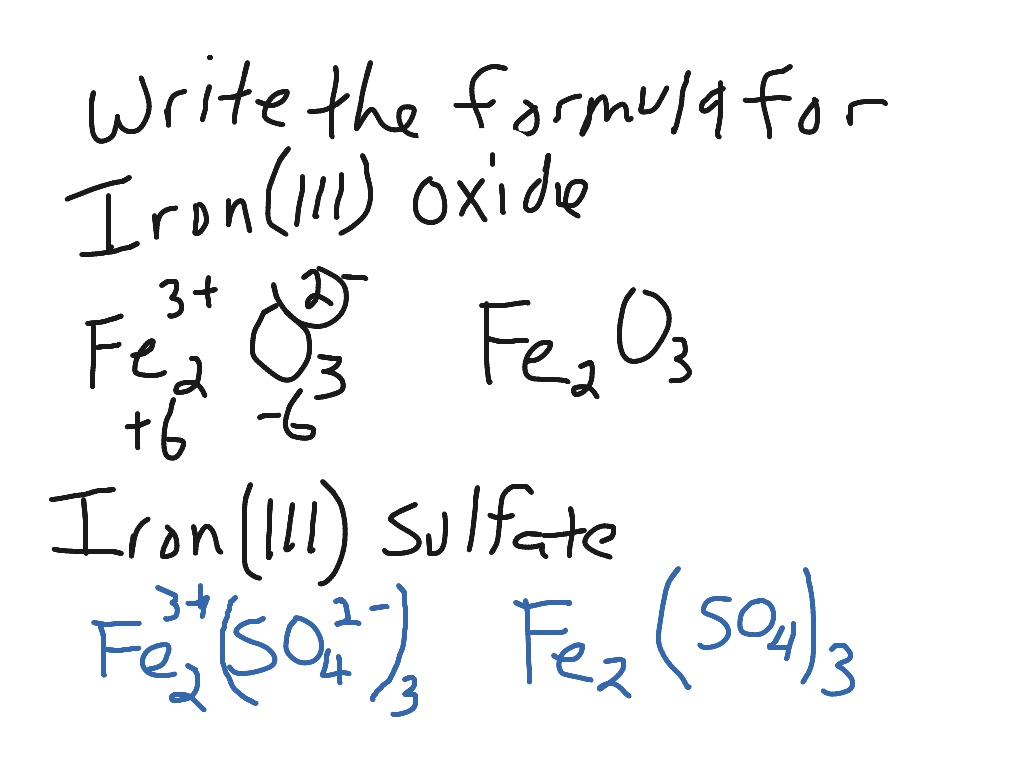Question Video: Deducing the Ionic Formula of an Ionic Compound Where Both Ions Have Greater-Than-One Charge | Nagwa

SOLVED: Iron reacts with oxygen from the atmosphere t0 produce iron (II) oxide; also known as rust (FezOs). What chemical species is oxidized in this reaction? What is the reducing #gent? 4.
Iron is produced by the reduction of iron (III) oxide using carbon monoxide. Fe2O3(s) + 3CO(g) 2Fe(s) + 3CO2(g). How much Fe is produced from 1 kg of Fe2O3? - Quora

Determine the molecular formula of an oxide of iron in which the mass per cent of iron and oxygen are 69.9 and 30.1 respectively.