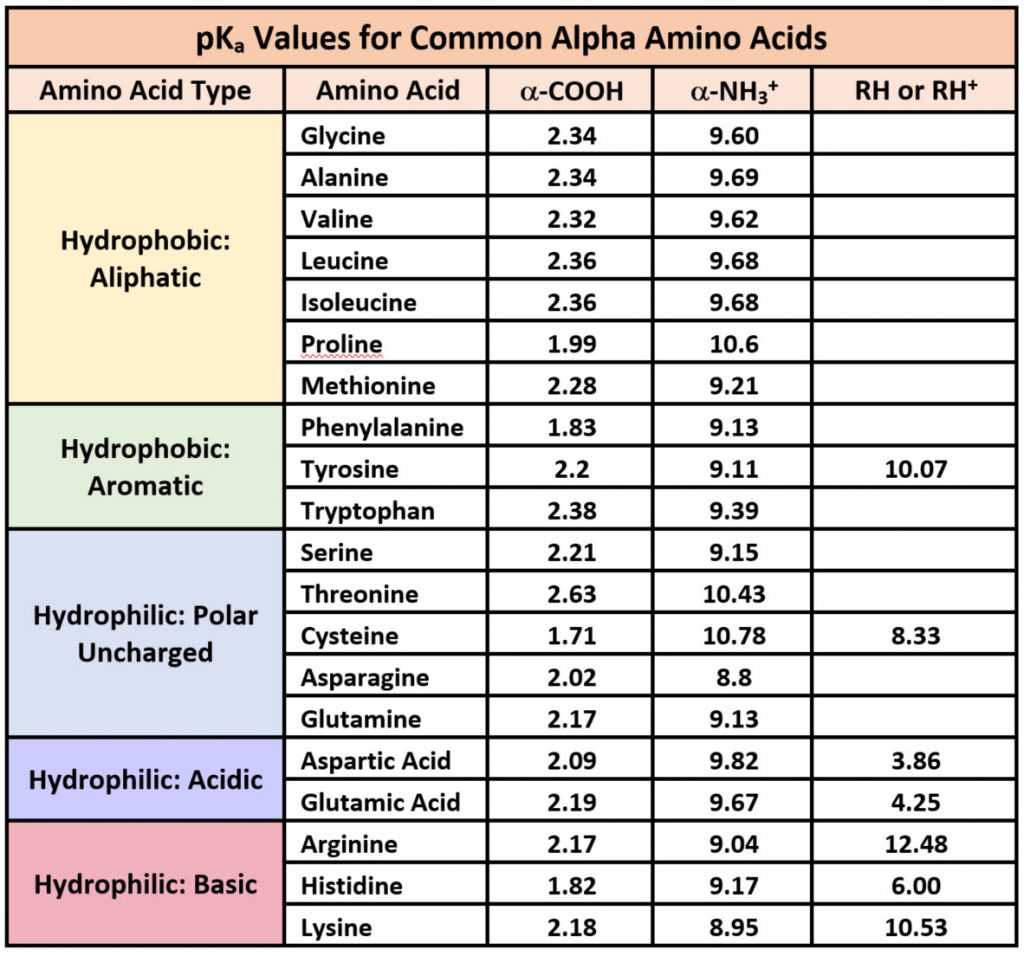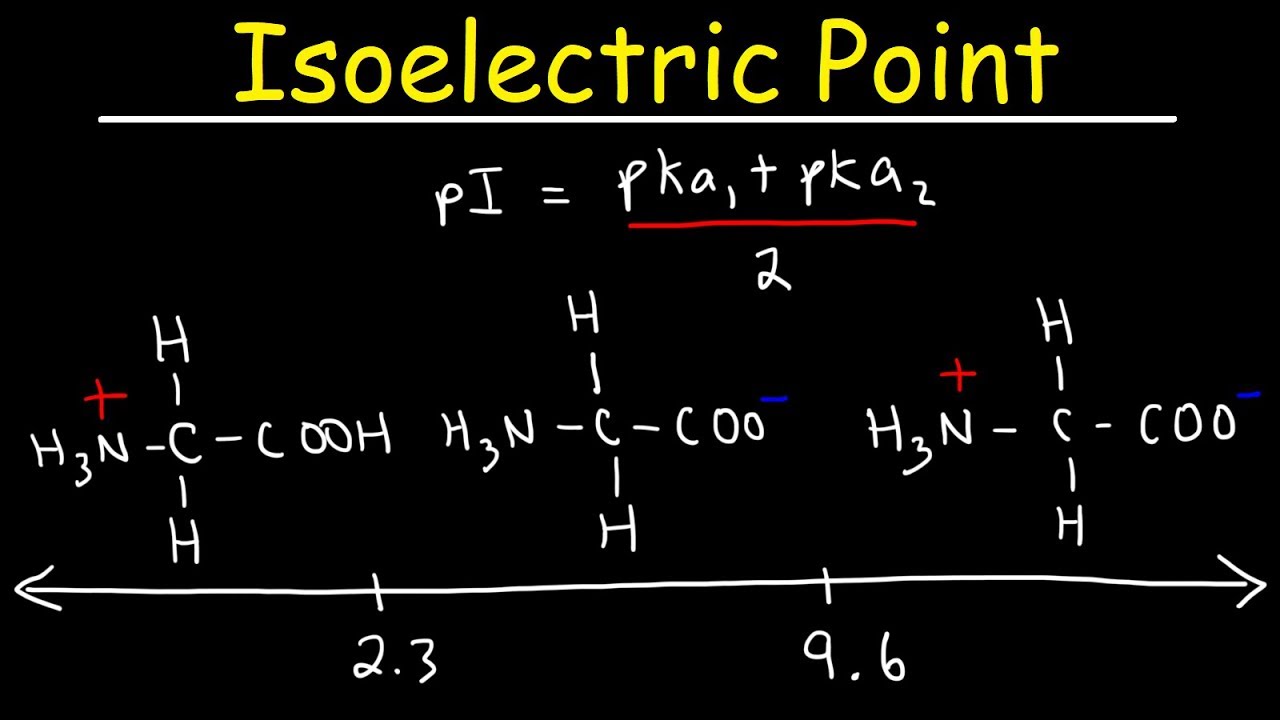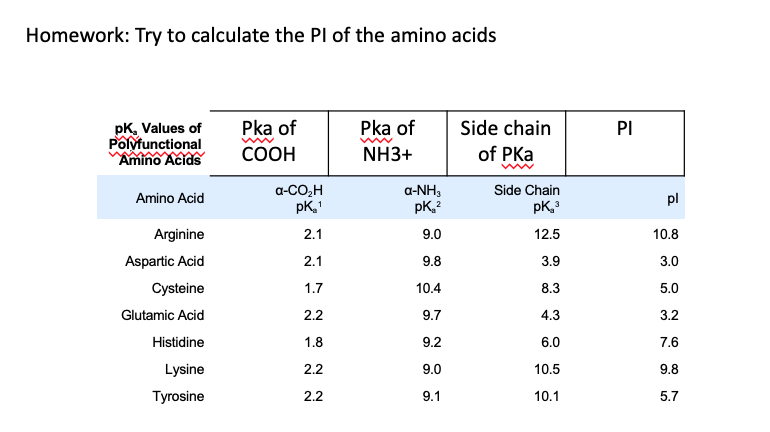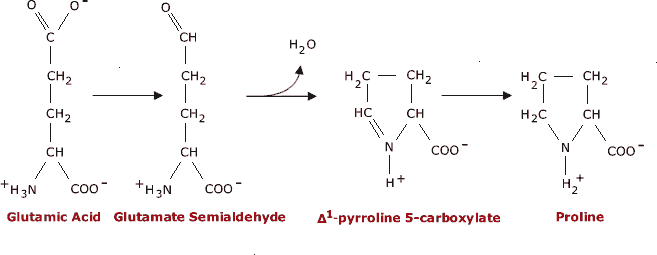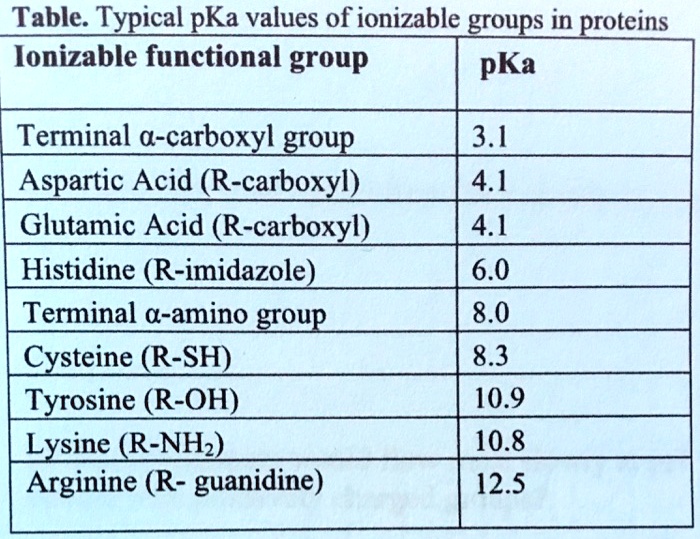
SOLVED: TableTypical pKa values of ionizable groups in proteins Ionizable functional group pKa Terminal a-carboxyl group Aspartic Acid (R-carboxyl) Glutamic Acid (R-carboxy)) Histidine (R-imidazole) Terminal @-aminogrOup Cysteine (R-SH) Tyrosine (R-OH ...

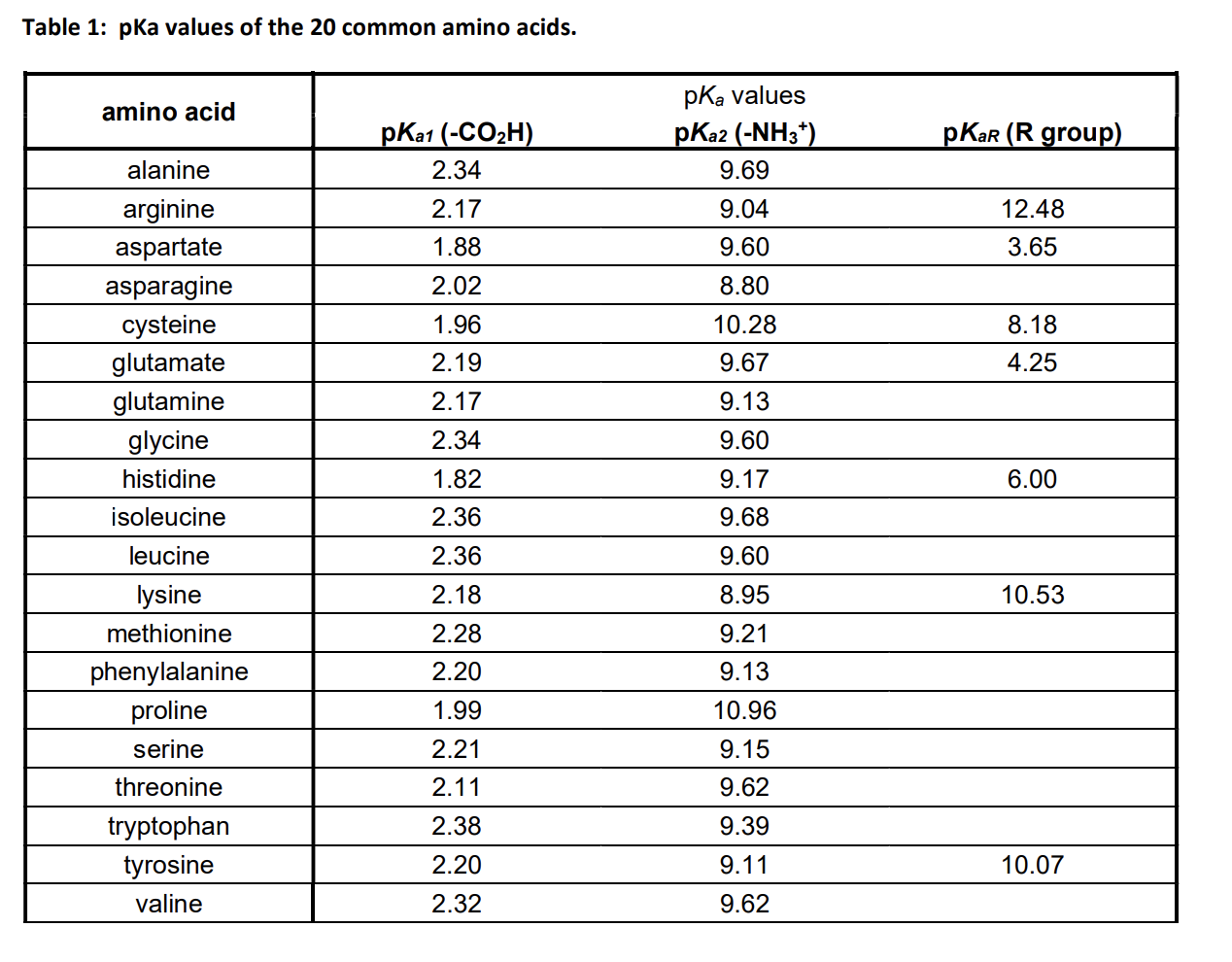
Structure of common basic and acidic amino acids, with the pKa values... | Download Scientific Diagram
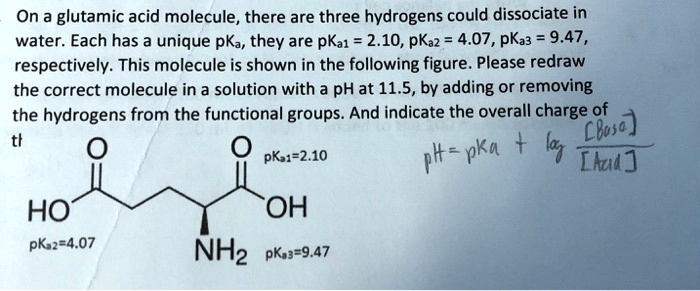
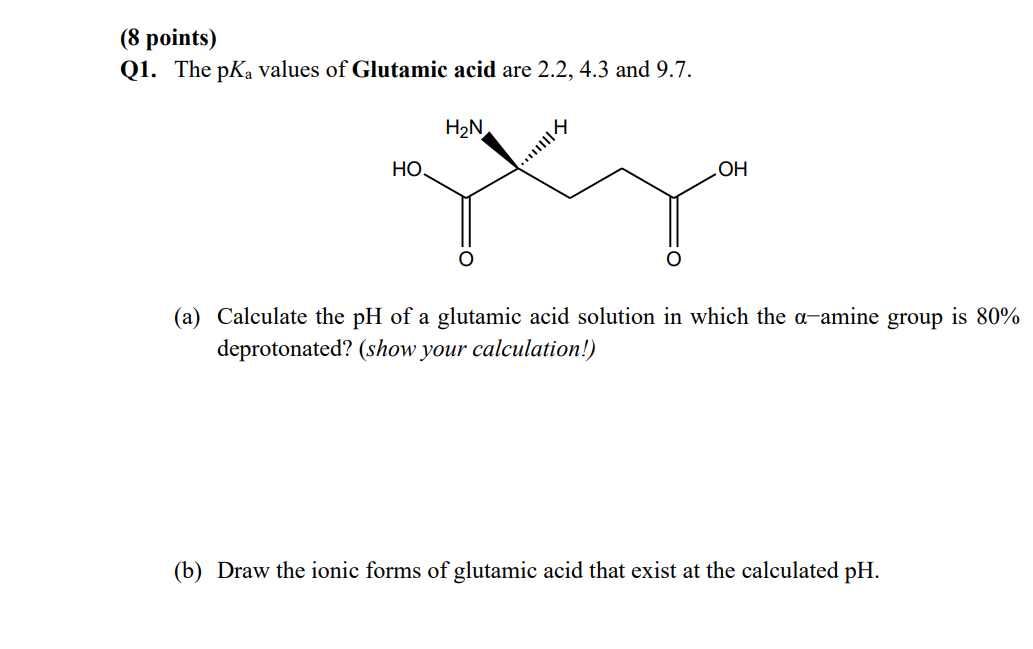
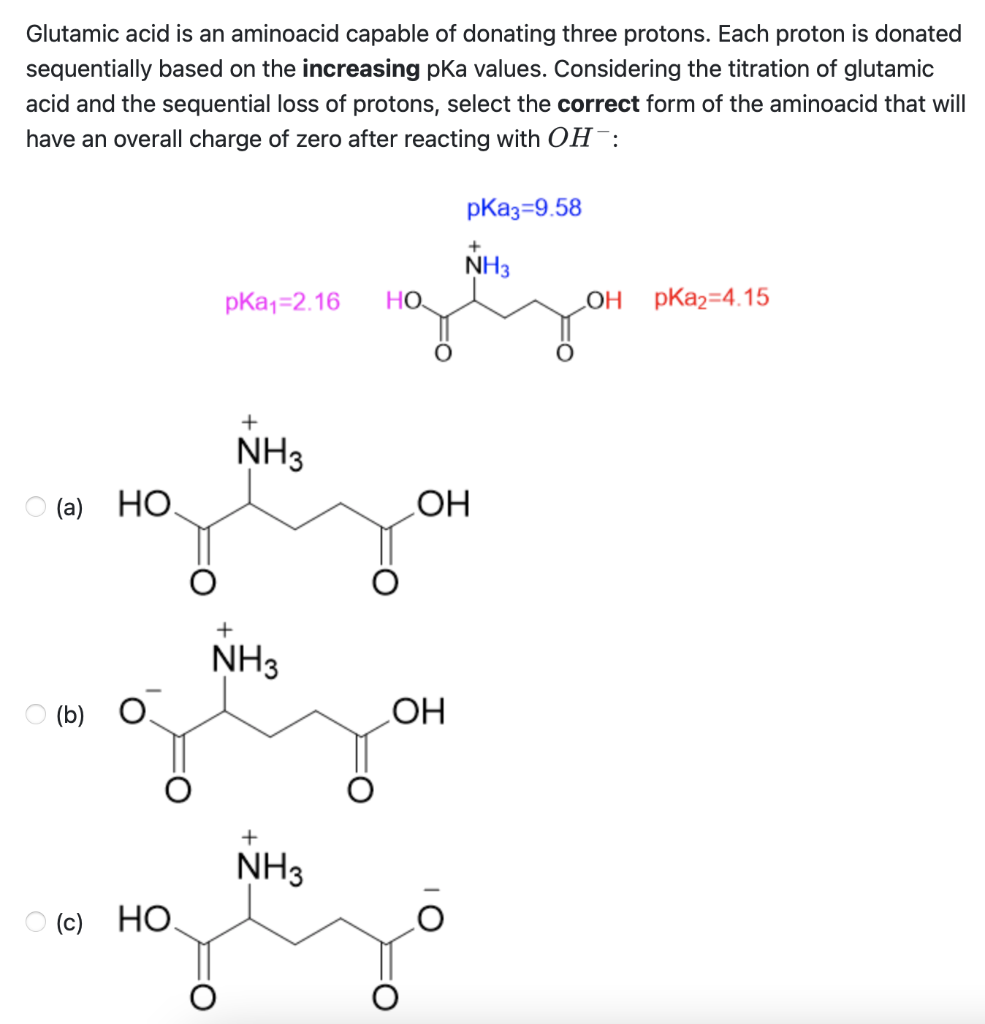
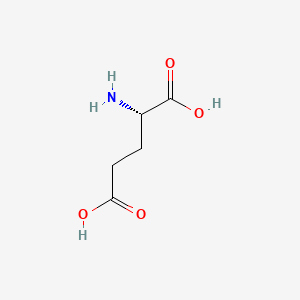
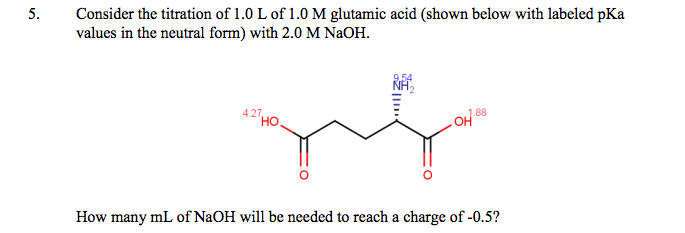
SOLVED: On a glutamic acid molecule, there are three hydrogens could dissociate in water. Each has unique pKa, they are pKa1 = 2.10, pKaz 4.07, pKaz = 9.47 respectively: This molecule is
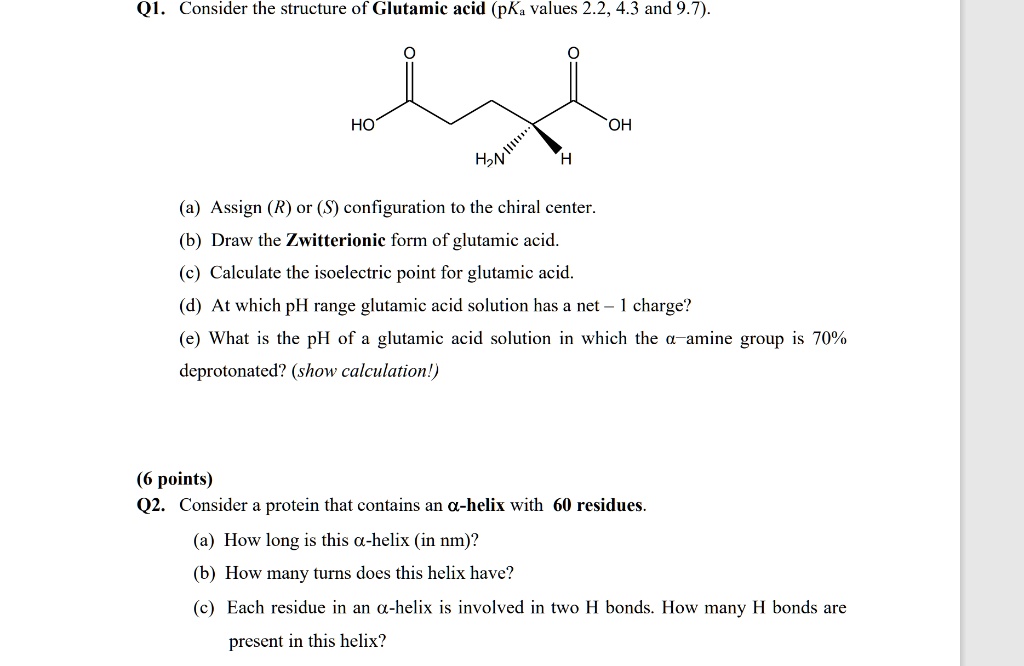
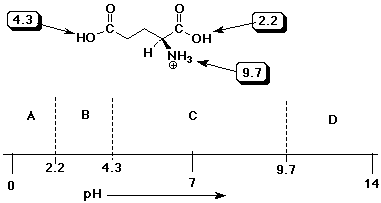
SOLVED: QI Consider the structure of Glutamic acid (pKa values 2.2, 4.3 and 9.7) HO OH Assign (R) or (S) configuration t0 the chiral center: (b) Draw the Zwitterionic form of glutamic