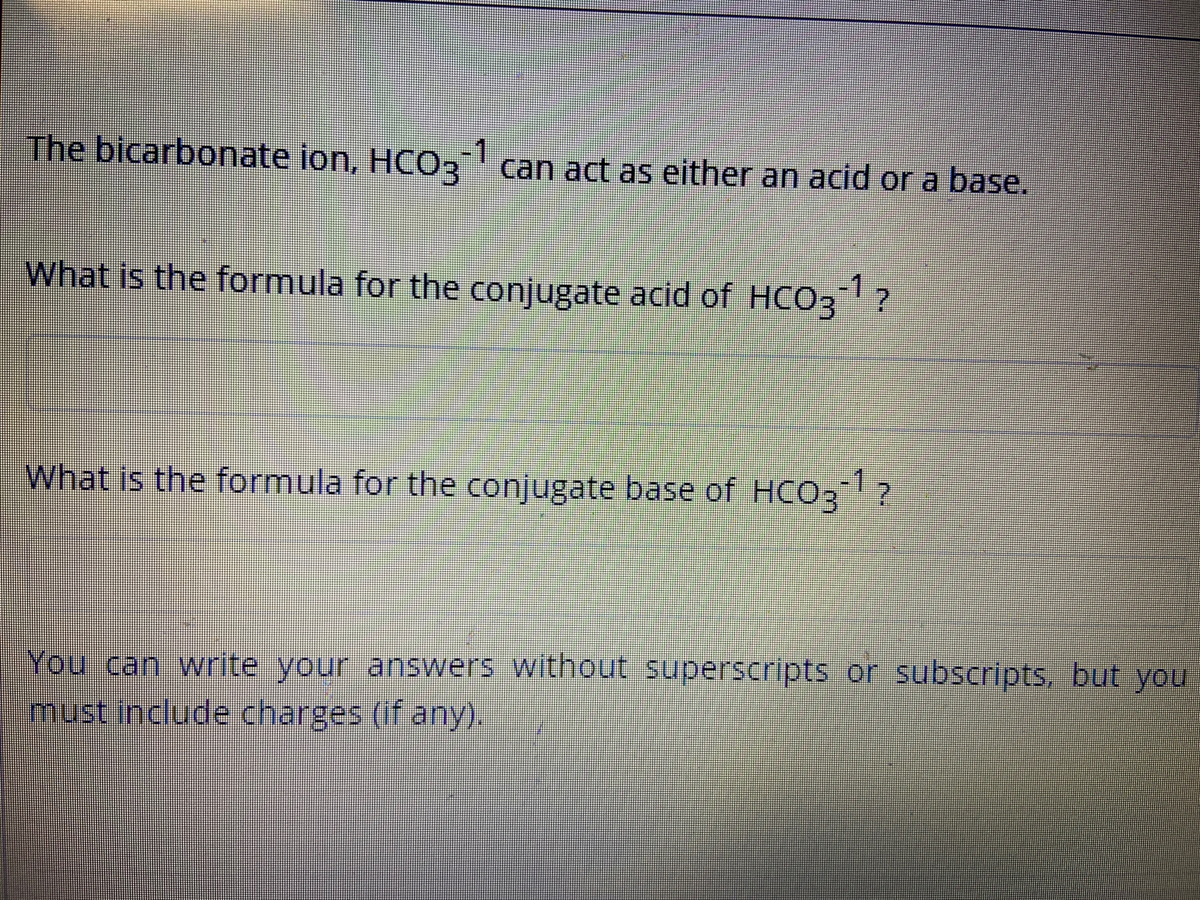
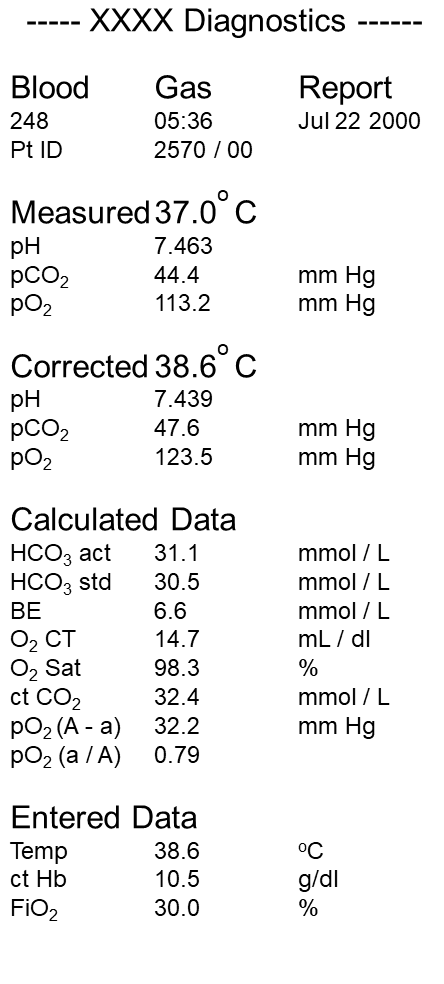
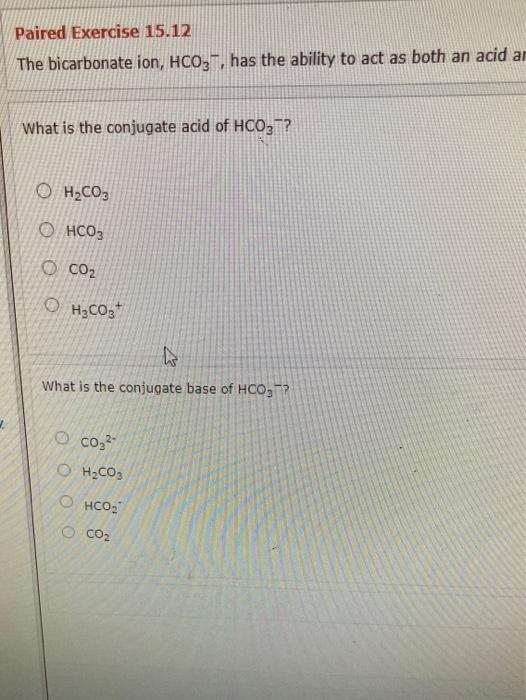
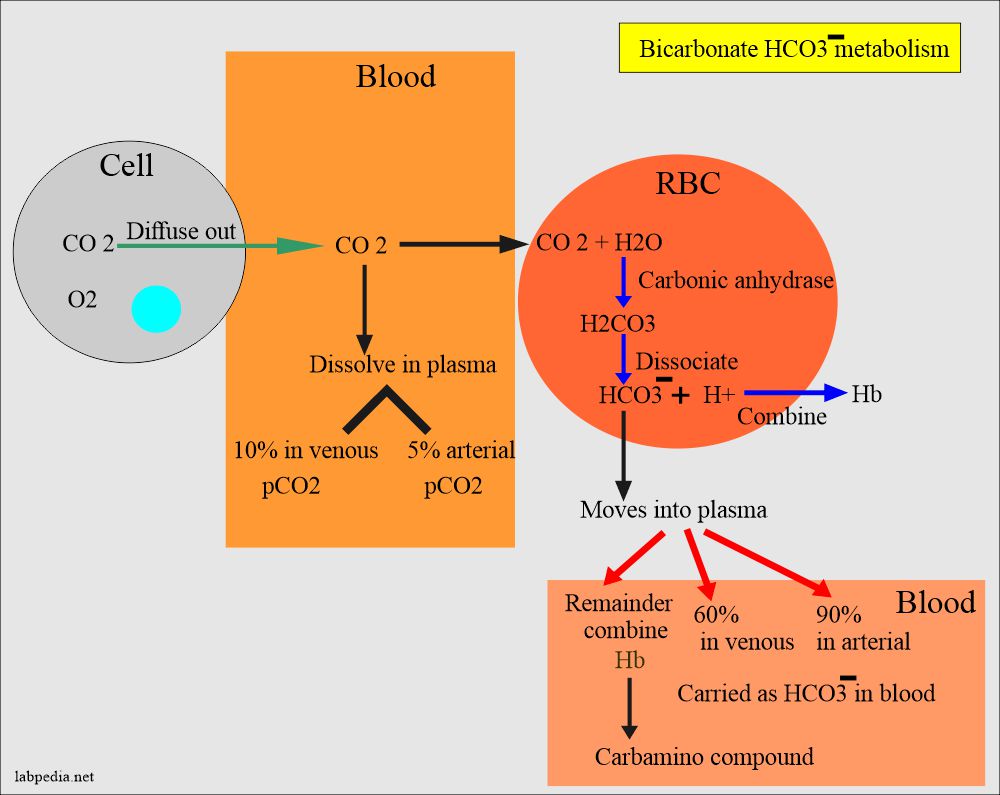
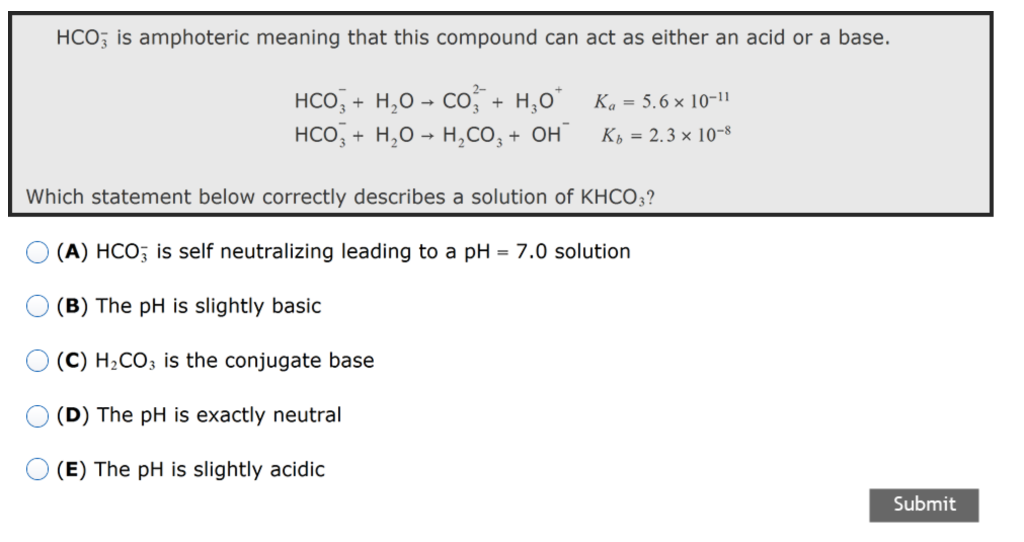
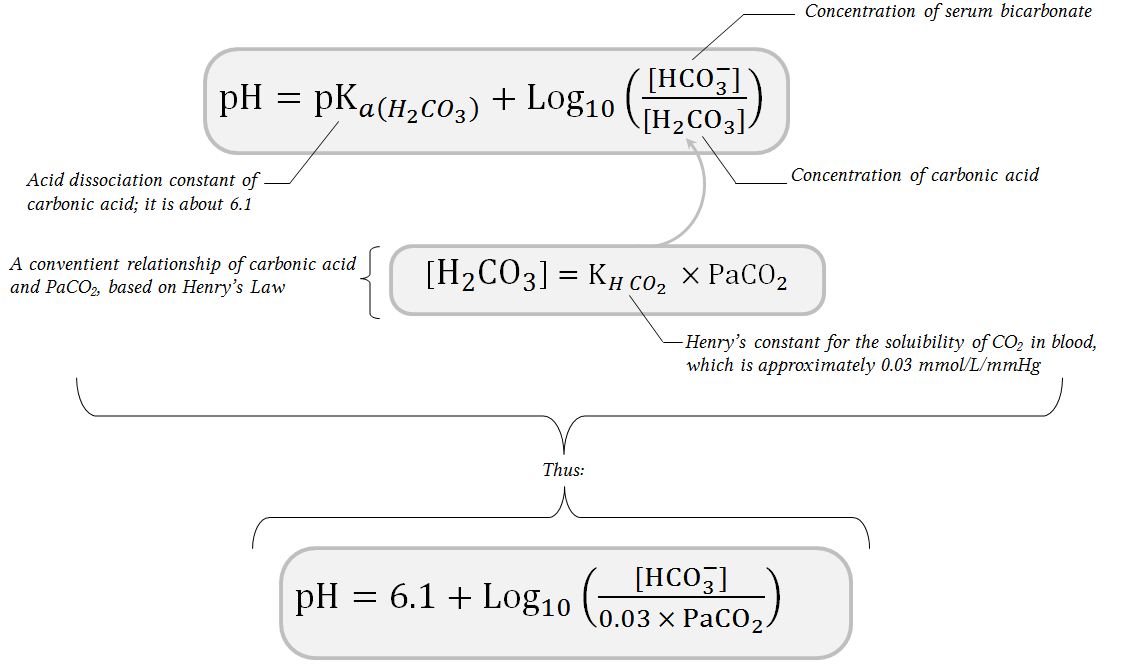
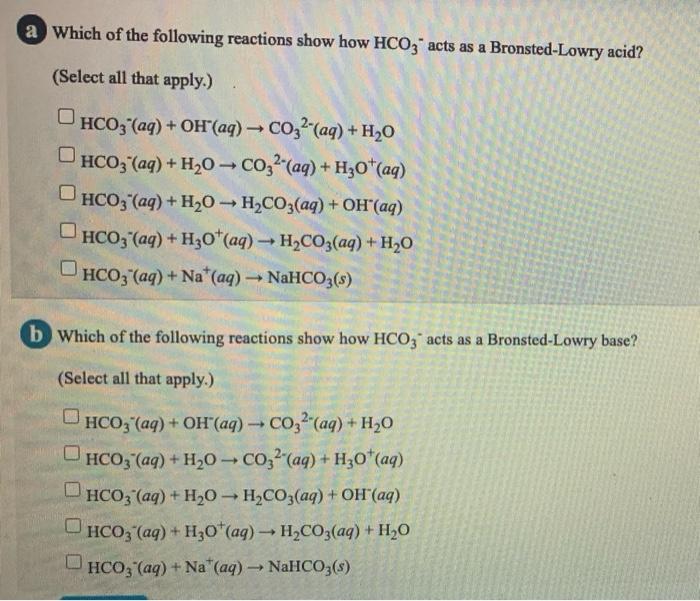SOLVED: Write an equation for HCO3- acting as an acid: HCOz" + Hzo <-> HzCO3 + OH- CO3 +H2o <-> HCOz- + OH- HCO3- <7> H+ + CO32- HCO:- +At <-7 HzCO3

The species: H2O, HCO3-, HSO4- and NH3 can act as Bronsted acids and bases. For each case give.... - YouTube