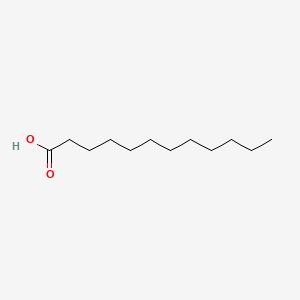
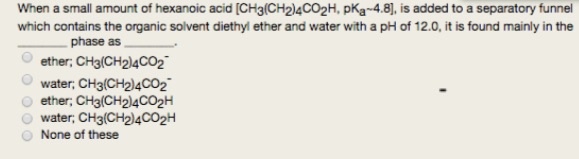Part A The pKa of cyclopentane is > 60, which is about what is expected for a hydrogen that is bonded to an sp3 carbon. Explain why cyclopentadiene is a much stronger

A Reliable and Efficient First Principles-Based Method for Predicting pKa Values. 2. Organic Acids | The Journal of Physical Chemistry A
![SOLVED: [ ] pKa of weak acids at 25*C Name Formula acetic acid CH3COzH benzoic acid C6HsCOzH butanoic acid CzHzCOzH 4-chlorobutanoic acid CzHsCICOzH crotonic acid CzHsCOzH oxalic acid HzC204 phosphoric acid HaPOa SOLVED: [ ] pKa of weak acids at 25*C Name Formula acetic acid CH3COzH benzoic acid C6HsCOzH butanoic acid CzHzCOzH 4-chlorobutanoic acid CzHsCICOzH crotonic acid CzHsCOzH oxalic acid HzC204 phosphoric acid HaPOa](https://cdn.numerade.com/ask_images/346c7f228e504822a839e28cc57b4b36.jpg)
SOLVED: [ ] pKa of weak acids at 25*C Name Formula acetic acid CH3COzH benzoic acid C6HsCOzH butanoic acid CzHzCOzH 4-chlorobutanoic acid CzHsCICOzH crotonic acid CzHsCOzH oxalic acid HzC204 phosphoric acid HaPOa

Using Atomic Charges to Describe the pKa of Carboxylic Acids | Journal of Chemical Information and Modeling

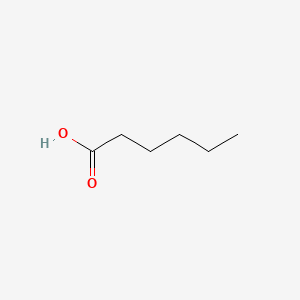
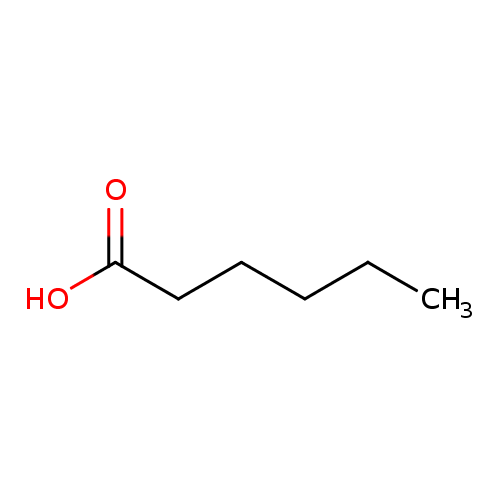
SOLVED: Dapointa Ching NA12 CM010 LcOien Your Hemenolnn In aqueous solution, hexanoic acid (HC6H1102) dissociates to produce its conjugate base, the hexanoate ion (C6H11Oz ), and the hydronium ion (H3o+) The solution
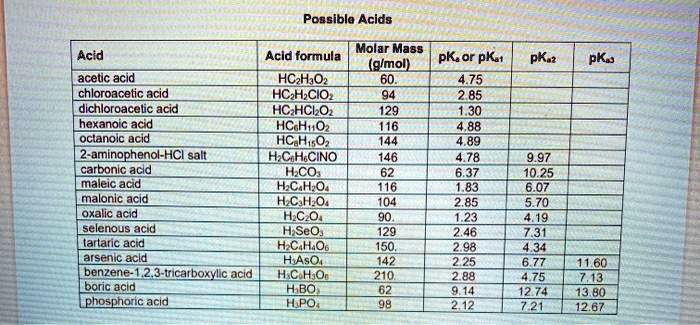
SOLVED: Poagibla Acids Acid Molar Mass Acid formula PK or pKt (gImol) PK ? PKa acelic acid HCzH,Oz chloroacetic acid HC H CIO dichloroacelic acid HC HCLO 129 hexanoic acid HCuOz oclanoic

Draw a flowchart that shows how you would seperate benzoic acid from 3,4-dibromophenol. Remember, both compounds are acidic. Look at pKa values to determine which is the most acidic. | Homework.Study.com
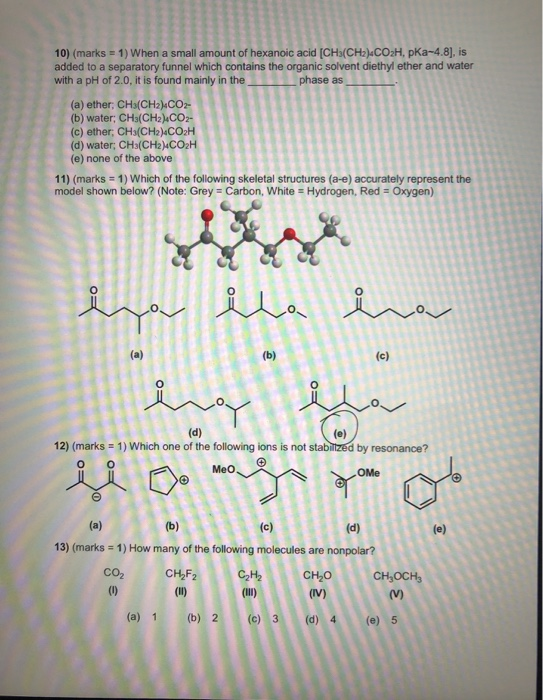
![OneClass: When a small amount of hexanoic acid [CH_3(CH_2)_4CO_2H, pKa ~ 4.8], is added to a separato... OneClass: When a small amount of hexanoic acid [CH_3(CH_2)_4CO_2H, pKa ~ 4.8], is added to a separato...](https://prealliance-textbook-qa.oneclass.com/qa_images/homework_help/question/qa_images/112/11211037.jpeg)