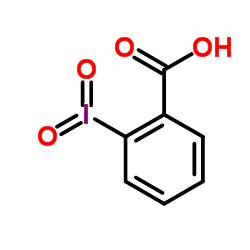
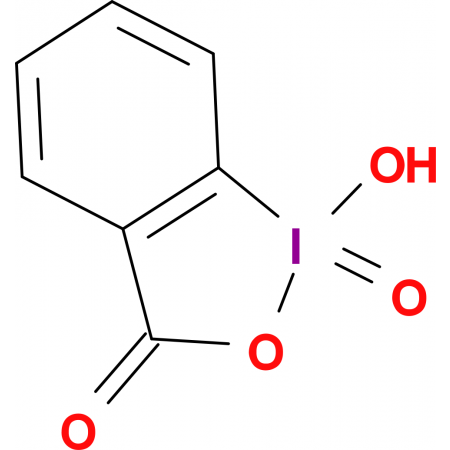
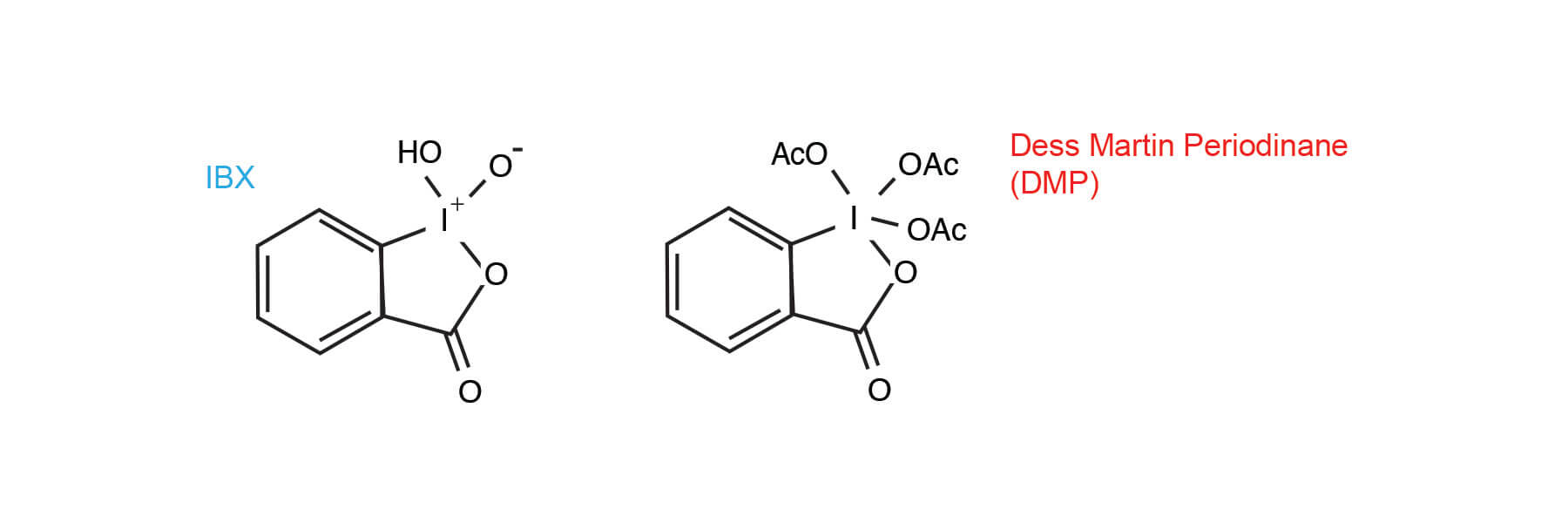
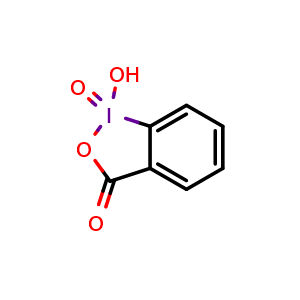
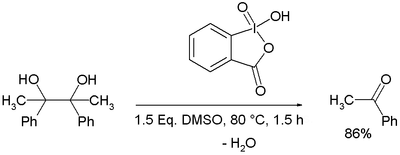
Hypervalent Iodine Oxidants: Structure and Kinetics of the Reactive Intermediates in the Oxidation of Alcohols and 1,2-Diols by o-Iodoxybenzoic Acid (IBX) and Dess−Martin Periodinane. A Comparative 1H-NMR Study | The Journal of

Caliente la venta el 99% CAS 61717-82-6 Ibx oxidante para Ácido Iodoxybenzoic 2 - China 2-Iodoxybenzoic ácido, 61717-82-6

o-Iodoxybenzoic Acid (IBX) as a Viable Reagent in the Manipulation of Nitrogen- and Sulfur-Containing Substrates: Scope, Generality, and Mechanism of IBX-Mediated Amine Oxidations and Dithiane Deprotections | Journal of the American Chemical

Modulation of the Reactivity Profile of IBX by Ligand Complexation: Ambient Temperature Dehydrogenation of Aldehydes and Ketones to α,β‐Unsaturated Carbonyl Compounds - Nicolaou - 2002 - Angewandte Chemie International Edition - Wiley Online Library

Hypervalent Iodine Oxidants: Structure and Kinetics of the Reactive Intermediates in the Oxidation of Alcohols and 1,2-Diols by o-Iodoxybenzoic Acid (IBX) and Dess−Martin Periodinane. A Comparative 1H-NMR Study | The Journal of

IBX-Mediated α-Hydroxylation of α-Alkynyl Carbonyl Systems. A Convenient Method for the Synthesis of Tertiary Alcohols | The Journal of Organic Chemistry

New Reactions of IBX: Oxidation of Nitrogen‐ and Sulfur‐Containing Substrates To Afford Useful Synthetic Intermediates - Nicolaou - 2003 - Angewandte Chemie - Wiley Online Library
![2,5-DICHLORO-3-(4,4,5,5-TETRAMETHYL-[1,3,2]-DIOXABOROLAN-2-YL)PYRIDINE CAS 1073371-98-8 100% factory price 2,5-DICHLORO-3-(4,4,5,5-TETRAMETHYL-[1,3,2]-DIOXABOROLAN-2-YL)PYRIDINE CAS 1073371-98-8 100% factory price](https://www.sincerechemical.com/wp-content/uploads/2022/11/25-DICHLORO-3-4455-TETRAMETHYL-132-DIOXABOROLAN-2-YLPYRIDINE-CAS-1073371-98-8.jpg)
2,5-DICHLORO-3-(4,4,5,5-TETRAMETHYL-[1,3,2]-DIOXABOROLAN-2-YL)PYRIDINE CAS 1073371-98-8 100% factory price

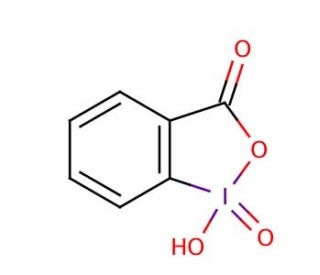
o‐Iodoxybenzoic Acid (IBX): pKa and Proton‐Affinity Analysis - Gallen - 2006 - Angewandte Chemie - Wiley Online Library

o-Iodoxybenzoic Acid (IBX) as a Viable Reagent in the Manipulation of Nitrogen- and Sulfur-Containing Substrates: Scope, Generality, and Mechanism of IBX-Mediated Amine Oxidations and Dithiane Deprotections | Journal of the American Chemical