![Write IUPAC names of the following coordination compounds.a) [Co(NH3)6]Cl3 b) K3[Fe(CN)6] c) K2[Pd(Cl)4] d) [Ni(Co)4] Write IUPAC names of the following coordination compounds.a) [Co(NH3)6]Cl3 b) K3[Fe(CN)6] c) K2[Pd(Cl)4] d) [Ni(Co)4]](https://i.ytimg.com/vi/VpeKIvrO0ZQ/maxresdefault.jpg)
Write IUPAC names of the following coordination compounds.a) [Co(NH3)6]Cl3 b) K3[Fe(CN)6] c) K2[Pd(Cl)4] d) [Ni(Co)4]
![Why Is [Ni(NH3)6]Cl2 Paramagnetic But [Co(NH3)6]Cl3 Is Diamagnetic ? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium Why Is [Ni(NH3)6]Cl2 Paramagnetic But [Co(NH3)6]Cl3 Is Diamagnetic ? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium](https://miro.medium.com/v2/resize:fit:1280/0*8wwL8Ru43LyLIIL8.png)
Why Is [Ni(NH3)6]Cl2 Paramagnetic But [Co(NH3)6]Cl3 Is Diamagnetic ? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium

Ions Atoms that lose electrons (negative) have more positive charge than negative charge so they are positive ions. Atoms that gain electrons (negative) - ppt download

The calculated molar absorbance spectra for nickel species (Ni 2+ ,... | Download Scientific Diagram
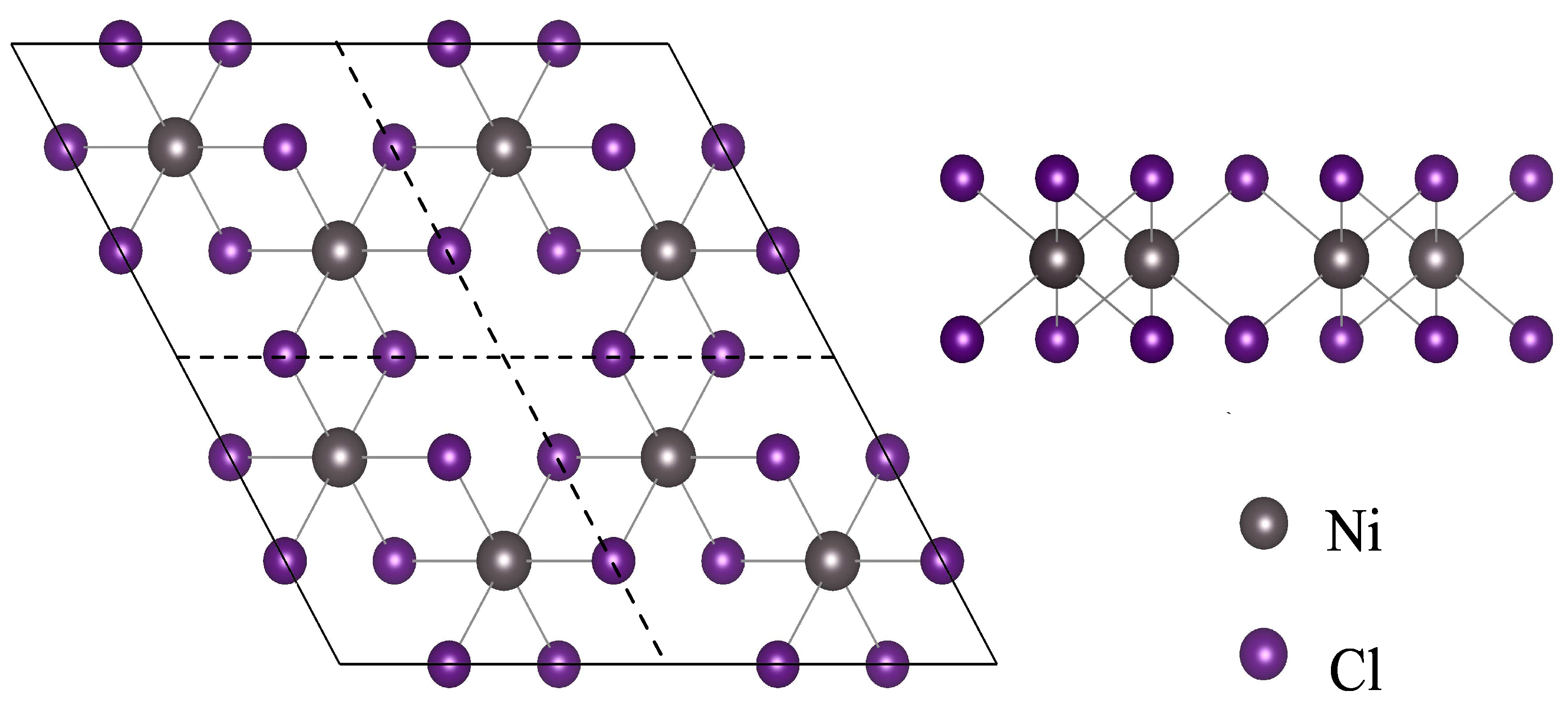
Nanomaterials | Free Full-Text | Thermoelectric Properties of NiCl3 Monolayer: A First-Principles-Based Transport Study
![Among the following, which complex compound is diamagnetic?i) [Co(NH3)6]Cl3 ,ii) [Ni(NH3)6]Cl2 ,iii) [Cr(H2O)6]Cl3 ,iv) [Fe(H2O)6]Cl2 Among the following, which complex compound is diamagnetic?i) [Co(NH3)6]Cl3 ,ii) [Ni(NH3)6]Cl2 ,iii) [Cr(H2O)6]Cl3 ,iv) [Fe(H2O)6]Cl2](https://d1hhj0t1vdqi7c.cloudfront.net/v1/V0FNUzRla0szUnM=/sd/)
Among the following, which complex compound is diamagnetic?i) [Co(NH3)6]Cl3 ,ii) [Ni(NH3)6]Cl2 ,iii) [Cr(H2O)6]Cl3 ,iv) [Fe(H2O)6]Cl2
![Scanning electron microscope (SEM) images of (a) [Fe(L)]Cl3 and (b)... | Download Scientific Diagram Scanning electron microscope (SEM) images of (a) [Fe(L)]Cl3 and (b)... | Download Scientific Diagram](https://www.researchgate.net/publication/356820649/figure/fig4/AS:1117440197033984@1643429912414/Scanning-electron-microscope-SEM-images-of-a-FeLCl3-and-b-NiLCl2-complexes.png)
Scanning electron microscope (SEM) images of (a) [Fe(L)]Cl3 and (b)... | Download Scientific Diagram
Among (i) [Co(NH3)6]Cl3, (ii) [Ni(NH3)6]Cl2, (iii) [Cr(H2O)6]Cl3, (iv) [Fe(H2O)6]Cl2 the complex which is diamagnetic is - Sarthaks eConnect | Largest Online Education Community
![FTIR spectra of a Ni(Sal)2, b [Cr(en)3]Cl3.3H2O, and c NiCr2O4 (sample 2) | Download Scientific Diagram FTIR spectra of a Ni(Sal)2, b [Cr(en)3]Cl3.3H2O, and c NiCr2O4 (sample 2) | Download Scientific Diagram](https://www.researchgate.net/publication/313686161/figure/fig1/AS:941596942024714@1601505613109/FTIR-spectra-of-a-NiSal2-b-Cren3Cl33H2O-and-c-NiCr2O4-sample-2.gif)
FTIR spectra of a Ni(Sal)2, b [Cr(en)3]Cl3.3H2O, and c NiCr2O4 (sample 2) | Download Scientific Diagram





![OneClass: What are all the isomers of [NiCl3(H2O)][PF6]? OneClass: What are all the isomers of [NiCl3(H2O)][PF6]?](https://prealliance-textbook-qa.oneclass.com/qa_images/homework_help/question/qa_images/121/12105789.png)



![Solved 5. Name the following compounds. a) [Cr(NH3)6]Cl3 b) | Chegg.com Solved 5. Name the following compounds. a) [Cr(NH3)6]Cl3 b) | Chegg.com](https://media.cheggcdn.com/media/a6d/s918x302/a6df084b-878c-469e-989e-bb7aab4ee9c3/image.png)