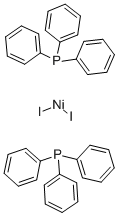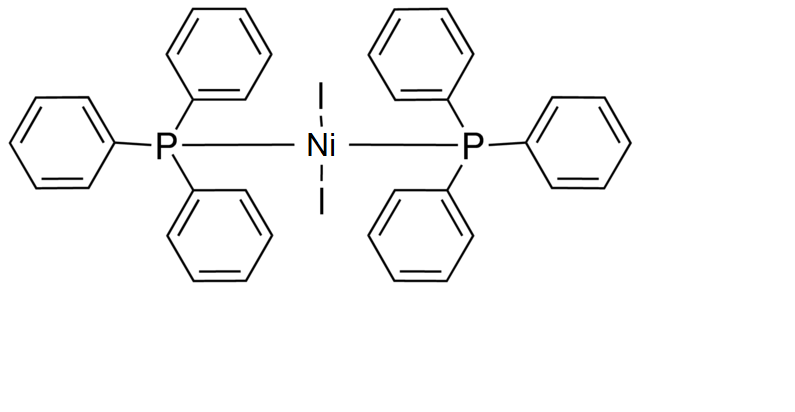
Diiodobis(triphenylphosphine)nickel(II) | Bis(triphenylphosphine)nickel(II) iodide | C36H30I2NiP2 - Ereztech
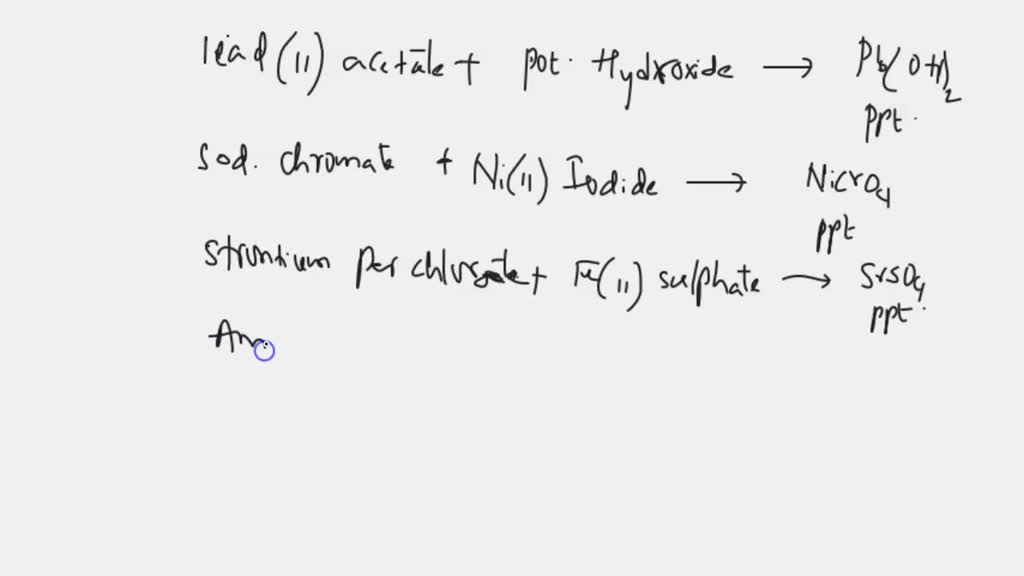
SOLVED: Write the formulas of the precipitates that would form when the following solutions are mixed, or write none if no precipitate would be produced. The Solubility Rules are needed to answer

SOLVED: Complete the table below for calculating the molar mass of the ionic compound nickel(II) iodide Number of ious Mass of ion in one mole of nickel(I) iodide Formula Molar mass of

SOLVED: so here are some of the questions i have please write clearly and answer them all, dont answer just one 1)Does a reaction occur when aqueous solutions of nickel(II) iodide and

Nickel(II) iodide, anhydrous, 99.5% (metals basis), Thermo Scientific Chemicals, Quantity: 5 g | Fisher Scientific

SOLVED: Tne names and chemical formulae of some chemical compounds are written in the Imagine that a few tenths of a mole of each compound is dissolved in a liter of wa