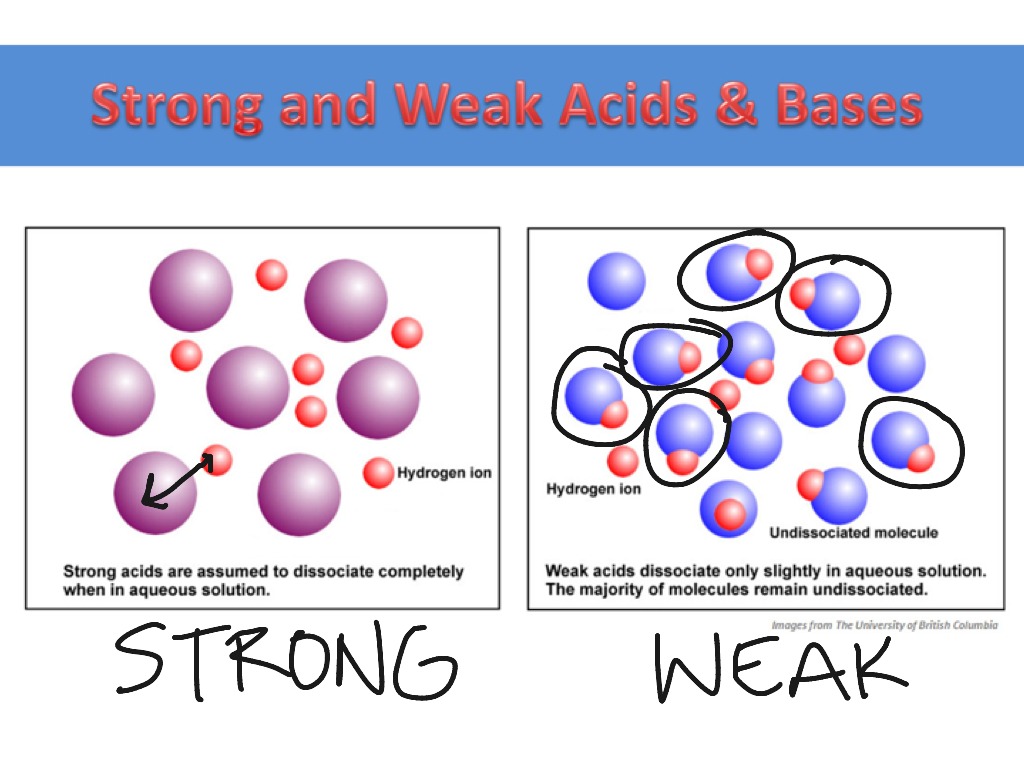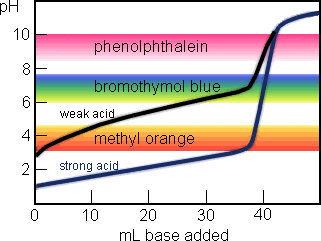
General Chemistry Online: FAQ: Acids and bases: How can strong and weak acids be distinguished using indicators?

Eurekly - 💪 STRONG AND WEAK ACIDS 💧 As opposed to STRONG acids, WEAK acids do not completely dissociate in water. Here they are: HO2C2O2H - oxalic acid H2SO3 - sulfurous acid
extraction - Why is a weak base more able to deprotonate a strong acid than a weak acid? - Chemistry Stack Exchange
![In the following list of acids, separate strong acids from weak acids :[Hydrochloric acid, Citric acid, Acetic acid, Nitric acid, Formic acid, Sulphuric acid.] In the following list of acids, separate strong acids from weak acids :[Hydrochloric acid, Citric acid, Acetic acid, Nitric acid, Formic acid, Sulphuric acid.]](https://dwes9vv9u0550.cloudfront.net/images/4730475/870f7400-e66d-4284-b36a-8a440dca9ba0.jpg)

:max_bytes(150000):strip_icc()/list-of-strong-and-weak-acids-603642-v2copy2-5b47abd0c9e77c001a395e55.png)







:max_bytes(150000):strip_icc()/list-of-strong-and-weak-acids-603642-v2copy2-5b47abd0c9e77c001a395e55.png)
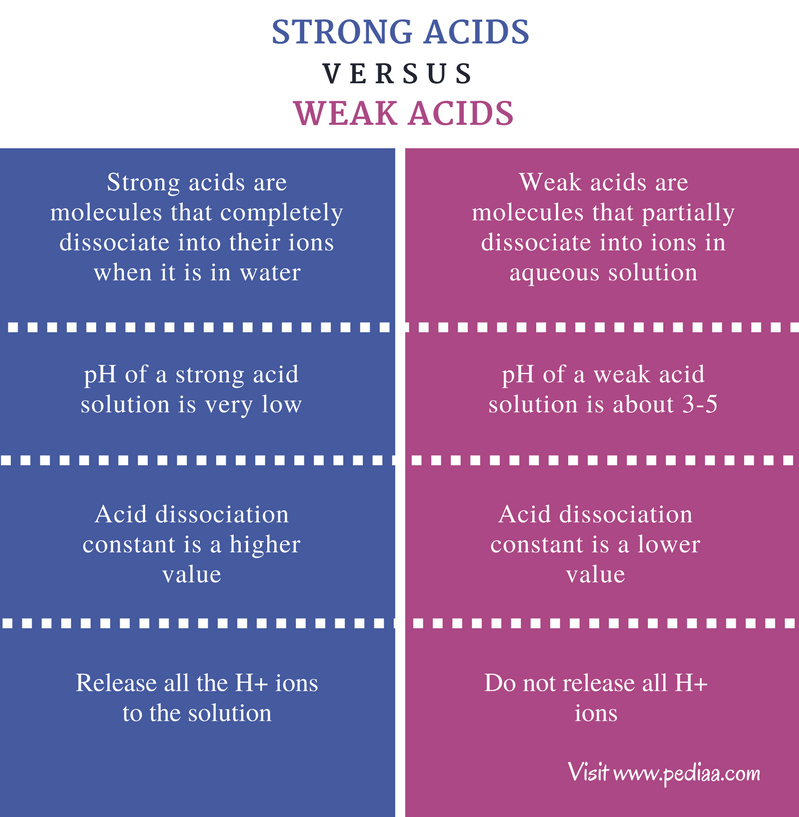
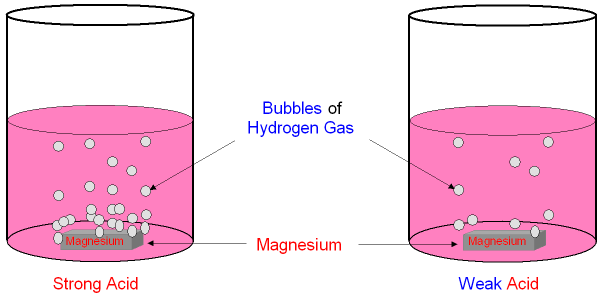
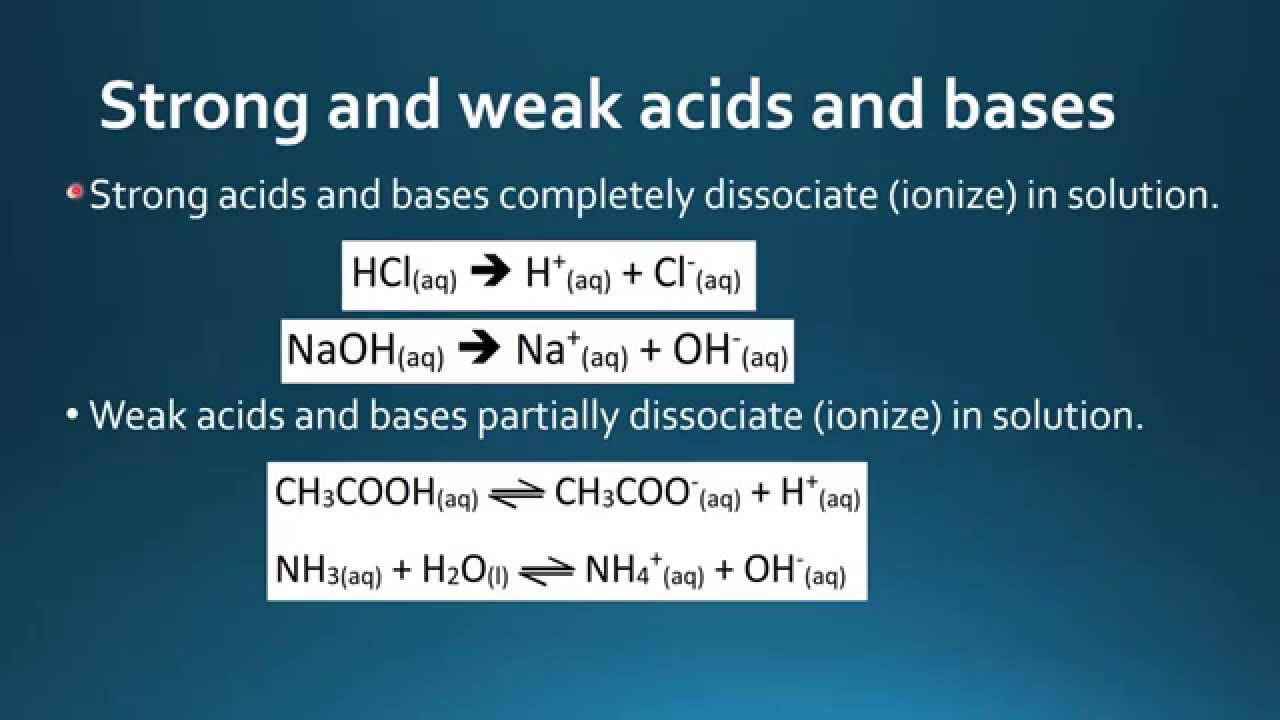
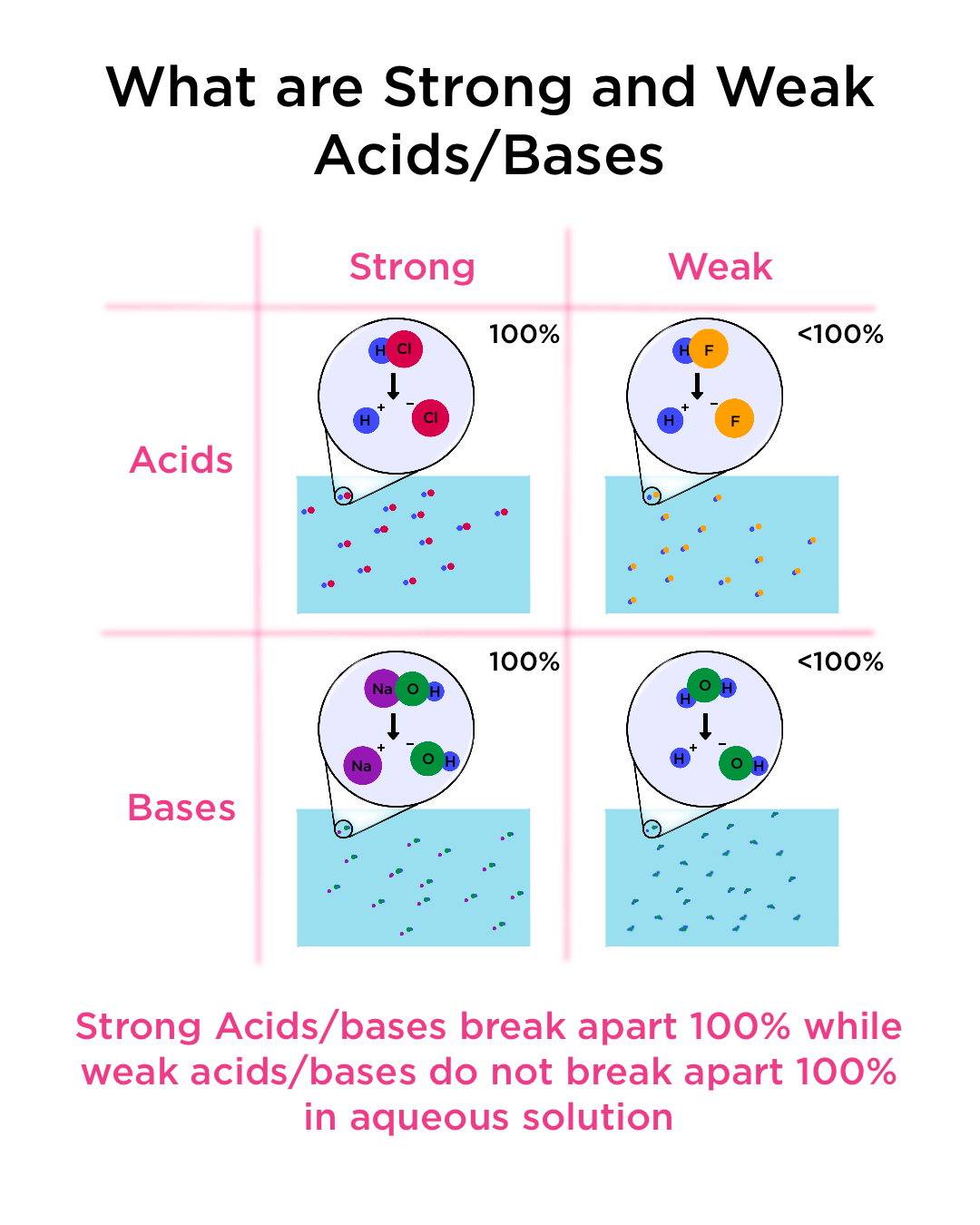
![Difference between Strong and Weak Acids [in Table Form] - Teachoo Difference between Strong and Weak Acids [in Table Form] - Teachoo](https://d1avenlh0i1xmr.cloudfront.net/eb34c57a-aeb2-4e7c-aa0b-613a11a6595e/differentiate-between-strong-and-weak-acids-01.jpg)